Specifications
- Purity
- ≥98% (HPLC)
- Appearance
- Yellow powder
- Identity
- 1H-NMR
Properties
- Solvents
- DMSO (10 mg/ml), DMF (10 mg/ml), ethanol (2 mg/ml)
Downloads
- Safety Data Sheet
- CDX F0336 MSDS.pdf
Category: Phytochemicals
One of the major polyphenolic flavonoids found in various fruits and vegetables such as apples, grapes, persimmons, strawberries, cucumbers and onions. Exerts numerous beneficial biological activities, including antibacterial, antifungal, antioxidant, anti-inflammatory, antiangiogenic, antiproliferative, proapoptotic, cell cycle arrest, autophagy inducing, hypolipidemic, neuroprotective and antitumor effects. Modulates multiple signaling pathways such as blocking of the phosphatidylinositol-3-kinase/protein kinase B/mammalian target of rapamycin (PI3K/Akt/mTOR) and p38, mitogen-activated protein kinases (MAPK)-dependent nuclear factor kappa-light-chain-enhancer of activated B cells (NF-?B) signaling, Wnt and adrogen receptor signaling, which playing a central role in various cellular processes contributing to the malignancy. Binds to β-tubulin and disrupts microtubule dynamics. Activator of sirtuin 1 and PPARs, inhibitor of DNA topoisomerase II (topo II), inhibitor of CDK6and GSK-3beta. Recently used as fluorescent molecular probe in sodium dodecyl sulfate (SDS) induced Sol-Gel transition.
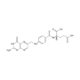
| Synonyms | 2-(3,4-dihydroxyphenyl)-3,7-dihydroxychromen-4-one, 3,3',4',7-Tetrahydroxyflavone, 5-Deoxyquercetin, Natural Brown 1, CI-75620, NSC 407010, NSC 656275, BRN 0292829, Cotinin, 528-48-3 (anhydrous) |
|---|---|
| Purity | ≥98% (HPLC) |
| Appearance | Yellow powder |
| CAS-Number | 528-48-3 |
| Molecular Formula | C15H10O6 |
| Molecular Weight | 286.24 |
| Identity | 1H-NMR |
| Solvents | DMSO (10 mg/ml), DMF (10 mg/ml), ethanol (2 mg/ml) |
| Smiles | OC1=CC=C2C(OC(C3=CC(O)=C(O)C=C3)=C(O)C2=O)=C1 |
| InChi Key | XHEFDIBZLJXQHF-UHFFFAOYSA-N |
| Shipping | AMBIENT |
| Short Term Storage | RT |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light and moisture. |
| Use / Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Transportation | Not dangerous goods |
| References | (1) M. Gabor & E. Eperjessy, Nature 212, 1273 (1966), (2) A.J. Olaharski, et al., Mutat. Res. 582, 79 (2005), (3) B. Sung, et al., Mol. Pharmacol. 71, 1703 (2007), (4) N. Khan, et al., Carcinogenesis 29, 1049 (2008), (5) B.G. Szczepankiewicz & P.Y. Ng, Curr. Top. Med. Chem. 8, 1533 (2008) (Review), (6) Y. Suh, et al., Carcinogenesis 30, 300 (2009), (7) Y. Suh, et al., Carcinogenesis 31, 1424 (2010), (8) R. Singh, et al., J. Med. Chem. 55, 3614 (2012), (9) V.M. Adhami, et al., Biochem. Pharmacol. 84, 1277 (2012) (Review), (10) D.N. Syed, et al., Anticancer Agents Med. Chem. 13, 995 (2013) (Review), (11) N. Khan, et al., Antioxid. Redox Signal. 19, 151 (2013) (Review), (12) C.H. Jung, et al., J. Nutr. Biochem. 24, 1547 (2013), (13) G.S. Prasath, et al., Eur. J. Pharmacol. 740, 248 (2014), (14) T. Jin, et al., J. Agric. Food Chem. 62, 10468 (2014), (15) E. Mukhtar, et al., Cancer Lett. 367, 173 (2015), (16) M.P. Reis, et al., J. Appl. Microbiol. 121, 373 (2016), (17) S.F. Nabavi, et al., Curr. Top. Med. Chem. 16, 1910 (2016) (Review), (18) T. Rengarajan & N.S. Yaacob, Eur. J. Pharmacol. 789, 8 (2016) (Review), (19) M. Youns, et al., PLoS One 12, e0169335 (2017), (20) J. Mishra, et al., J. Phys. Chem. B 122, 181 (2018), (21) K D. Kashyap, et al., Life Sci. 194, 75 (2018) (Review), (22) K. Shanmugam, et al., Oxid. Med. Cell Longev. 2018, 9173436 (2018) |
| InChi | InChI=1S/C15H10O6/c16-8-2-3-9-12(6-8)21-15(14(20)13(9)19)7-1-4-10(17)11(18)5-7/h1-6,16-18,20H |
| Quantity | 100 mg, 500 mg, Bulk |
 L-Glutathione oxidized
L-Glutathione oxidized