Specifications
- Purity
- ≥98% (HPLC)
- Appearance
- White to off-white powder
- Identity
- 1H-NMR
Properties
- Solvents
- water (15 mg/ml), DMSO, ethanol (10 mg/ml)
Category: Phytochemicals
Lycorine is an active alkaloid abundant in Amaryllidaceae with a wide range of biological properties, including antifungal, antiparasitic, antiviral, anti-inflammatory and anticancer activity. It is a protein synthesis by preventing amino acid incorporation into proteins, potentially through the inhibition of peptidyltransferases. Lycorine induces cell cycle arrest and cell death in Trichomonas. As anticancer agent on a molecular basis it induces G0/G1 phase cell cycle arrest and apoptosis, inhibits activity of histone deacetylases (HDACs), upregulates expression of p53 and p21, downregulates expression of cyclin D1 and CDK4, inhibits the Wnt/β-catenin pathway, activates ROCK1and inhibits JAK/STAT pathways. Lycorine exhibits antiviral activities against enterovirus, flaviviruses, Zika virus, HIV-1, SARS-CoV, SARS-CoV-2 and hepatitis C virus, most probably by suppressing viral replication. Lycorine has been shown to inhibit NLRP3 inflammasome activation and pyroptosis.
| Synonyms | Lycorine chloride |
|---|---|
| Purity | ≥98% (HPLC) |
| Appearance | White to off-white powder |
| CAS-Number | 2188-68-3 |
| Molecular Formula | C16H17NO4 . HCl |
| Molecular Weight | 323.77 |
| Identity | 1H-NMR |
| Solvents | water (15 mg/ml), DMSO, ethanol (10 mg/ml) |
| Smiles | O[C@H]([C@H]1O)C=C2CCN3CC4=CC5=C(OCO5)C=C4[C@@]1([H])[C@]32[H].Cl |
| InChi Key | VUVNTYCHKZBOMV-NVJKKXITSA-N |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use / Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Hazard statements | H301 |
| Precautionary statements | P301 + P310 |
| GHS Symbol | GHS06 |
| Signal word | Danger |
| RIDADR | UN2811 |
| Transportation | Packing Group III |
| References | (1) M. Kukhanova, et al., FEBS Lett. 160, 129 (1983), (2) R.B. Giordani, et al., Phytochemistry 72, 645 (2011), (3) L. Li, et al., Cancer Cell Int. 12, 49 (2012), (4) Z. Cao, et al., Toxicol. Lett. 218, 174 (2013), (5) J.W. Shen, et al., J. Microbiol. Biotechnol. 24, 354 (2014), (6) S. Wu, et al., Front. Pharmacol. 9, 881 (2018), (7) M. Roy, et al., Biomed. Pharmacother. 107, 615 (2018), (8) W.Y. Liu, et al., Front. Pharmacol. 10, 651 (2019), (9) H. Hu, et al., Onco. Targets Ther. 12, 5377 (2019), (10) H. Wang, et al., Br. J. Haematol. 189, 1151 (2020), (11) Q. Liang, et al., Pharmacol. Res. 158, 104884 (2020), (12) H. Chen, et al., Virology 546, 88 (2020), (13) Y.N. Zhang, et al., Emerg. Microbes Infect. 9, 1170 (2020) |
| InChi | InChI=1S/C16H17NO4.ClH/c18-11-3-8-1-2-17-6-9-4-12-13(21-7-20-12)5-10(9)14(15(8)17)16(11)19,/h3-5,11,14-16,18-19H,1-2,6-7H2,1H/t11-,14-,15+,16+,/m0./s1 |
| Quantity | 20 mg, Bulk |

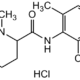 Mepivacaine hydrochloride
Mepivacaine hydrochloride