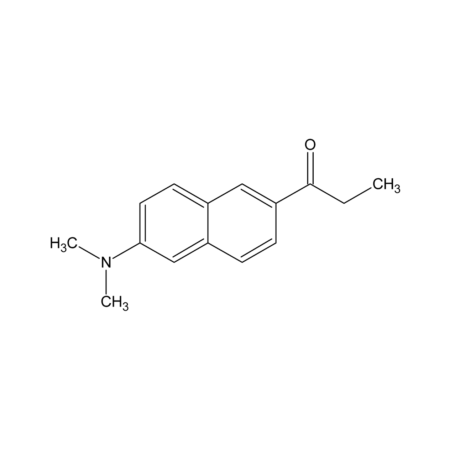
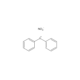
N,N-Dimethyl-6-propionyl-2-naphthylamine
- SKU
- D0077
Category: Fluorescent Detection, Fluorescent Detection
- Synonyms
- Prodan
- 70504-01-7
- CAS-Number
- C15H17NO
- Molecular Formula
- 227.3
- Molecular Weight
Specifications
- Purity
- ≥98% (HPLC)
- Appearance
- Light yellow powder
- Identity
- 1H-NMR
Properties
- Solvents
- methanol, DMF, acetonitrile or acetone
- Melting Point
- 137 °C
- Fluorescence
- λex 361 nm, λem 498 nm in methanol
Downloads
- Safety Data Sheet
- CDX D0077 MSDS.pdf
- Shipping
- AMBIENT
- Short Term Storage
- +4°C
- Long Term Storage
- -20°C
- Handling Advice
- Protect from light and moisture.
- Use / Stability
- Stable for at least 2 years after receipt when stored at -20°C.
- Transportation
- Not dangerous goods
- Description
- N,N-dimethyl-6-propionyl-2-naphthylamine (prodan) has both an electron-donor and an electron-acceptor substituent, resulting in a large excited-state dipole moment and extensive solvent polarity-dependent fluorescence shifts. When prodan is incorporated into membranes, their fluorescence spectra are sensitive to the physical state of the surrounding phospholipids. In membranes, prodan appears to localize at the surface, although Fourier transform infrared measurements indicate some degree of penetration into the lipid interior. Excited-state relaxation of prodan is sensitive to the nature of the linkage between phospholipid hydrocarbon tails and the glycerol backbone. Tubulin and its hydrophobic surfaces have been probed with the enviroment-sensitive probes prodan. Prodan is also used for the generation of peroxy-oxalate chemiluminescence with H2O2.
- Smiles
- CCC(=O)C1=CC2=CC=C(C=C2C=C1)N(C)C
- InChi Key
- MPPQGYCZBNURDG-UHFFFAOYSA-N
- References
- (1) B.A. Rowe et al.: J. Phys. Chem. B 110(30), 15021 (2006) , (2) E. Omanovic et al.: Int. J. Environm. Anal. Chem. 85(12-13), 853 (2005) , (3) M. Lin et al.: C. Huie: Anal. Chim. Acta 339(1-2), 131 (1997) , (4) A. Chakrabarti et al.: Biochem. Biophys. Res. Commun. 226, 495 (1996) , (5) P.L.G. Chong et al.: Biochemistry 28, 8358 (1989) , (6) P.L.G. Chong et al.: Biochemistry 27, 399 (1988)
- InChi
- InChI=1S/C15H17NO/c1-4-15(17)13-6-5-12-10-14(16(2)3)8-7-11(12)9-13/h5-10H,4H2,1-3H3
N,N-dimethyl-6-propionyl-2-naphthylamine (prodan) has both an electron-donor and an electron-acceptor substituent, resulting in a large excited-state dipole moment and extensive solvent polarity-dependent fluorescence shifts. When prodan is incorporated into membranes, their fluorescence spectra are sensitive to the physical state of the surrounding phospholipids. In membranes, prodan appears to localize at the surface, although Fourier transform infrared measurements indicate some degree of penetration into the lipid interior. Excited-state relaxation of prodan is sensitive to the nature of the linkage between phospholipid hydrocarbon tails and the glycerol backbone. Tubulin and its hydrophobic surfaces have been probed with the enviroment-sensitive probes prodan. Prodan is also used for the generation of peroxy-oxalate chemiluminescence with H2O2.