Specifications
- Purity
- ≥98% (HPLC)
- Appearance
- White to off-white powder
- Identity
- 1H-NMR
Properties
- Solvents
- ethanol (30 mg/ml), DMSO (15 mg/ml) or DMF (20 mg/ml).
- Melting Point
- 214 °C (decomp.)
- Density
- 1.329 (predicted)
Category: Metabolites
p-Coumaric acid is mainly a plant metabolite which exhibits antioxidant, antimicrobial, antitumor, antiangiogenic, antidiabetic, antihyperlipidemic, neuroprotective and anti-inflammatory properties. It shows bactericidal/quorum sensing activity by damaging bacterial cell membrane and by interacting with bacterial DNA. It prevents metabolic syndromes through the activation of brown adipose tissue (BAT), linked to UCP1 upregulation, with accelerated burning of fatty acids and glucose, leading to enhanced the energy expenditure and thermogenesis. Also used in proteomics, e.g. in western blot, p-coumaric is used as an enhancer in chemiluminescent substrates. Used in cosmetics as a skin-lightening compound, due to its antimelanogenic effects, based on its potent competitive inhibition of tyrosinase.
| Synonyms | para-Coumaric Acid, trans-p-Coumaric Acid, trans-4-Hydroxycinnamic acid, (E)-3-(4-Hydroxyphenyl)prop-2-enoic acid |
|---|---|
| Purity | ≥98% (HPLC) |
| Appearance | White to off-white powder |
| CAS-Number | 501-98-4 |
| Molecular Formula | C9H8O3 |
| Molecular Weight | 164.16 |
| Identity | 1H-NMR |
| Solvents | ethanol (30 mg/ml), DMSO (15 mg/ml) or DMF (20 mg/ml). |
| Melting Point | 214 °C (decomp.) |
| Density | 1.329 (predicted) |
| Smiles | OC1=CC=C(/C=C/C(O)=O)C=C1 |
| InChi Key | NGSWKAQJJWESNS-ZZXKWVIFSA-N |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use / Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Hazard statements | H315 – H319 – H335 |
| Precautionary statements | P302 + P352 – P305 + P351 + P338 |
| GHS Symbol | GHS07 |
| Transportation | Not dangerous goods |
| References | (1) C. Luceri, et al., Br. J. Nutr. 97, 458 (2007), (2) C. Haan & I. Behrmann, J. Immunol. Meth. 318, 11 (2007), (3) S.F. Bodini, et al., Lett. Appl. Microbiol. 49, 551 (2009), (4) D. Dolores & C.Y. Cheng, Spermatogenesis 1, 121 (2011), (5) D. Stojkovic, et al., J. Sci. Food Agric. 93, 3205 (2013), (6) S.J. Pragasam & M. Rasool, Inflamm. Res. 62, 489 (2013), (7) S.J. Pragasam, et al., Inflammation 36, 169 (2013), (8) C.S. Kong, et al., Phytother. Res. 27, 317 (2013), (9) S. Shailasree, et al., Mol. Neurobiol. 51, 119 (2015), (10) V. Amalan, et al., Biomed. Pharmacother. 84, 230 (2016), (11) K. Pei, et al., J. Sci. Food Agric. 96, 2952 (2016) (Review), (12) A. Abdel-Moneim, et al., Biomed. Pharmacother. 105, 1091 (2018), (13) S.H. Sharma, et al., Chem. Biol. Interact. 291, 16 (2018), (14) R. Sakamula, et al., Metab. Brain Dis. 33, 765 (2018), (15) Y.C. Boo, Antioxidants 8, 275 (2019) (Review), (16) X. Hu, et al., Front. Oncol. 10, 558414 (2020), (17) X. Han, et al., FASEB J. 34, 7810 (2020), (18) M.F. Abazari, et al., Mini Rev. Med. Chem. (epub ahead of print) (2021) (Review) |
| InChi | InChI=1S/C9H8O3/c10-8-4-1-7(2-5-8)3-6-9(11)12/h1-6,10H,(H,11,12)/b6-3+ |
| Quantity | 10 g, 25 g, Bulk |
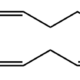 Eicosapentaenoic acid
Eicosapentaenoic acid