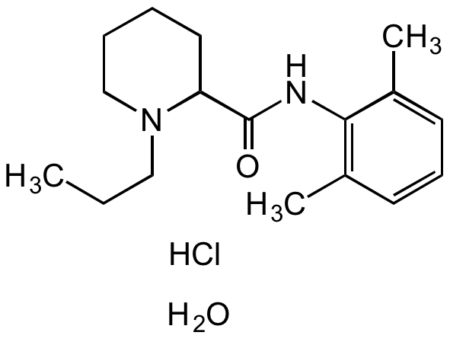
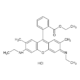
Ropivacaine hydrochloride monohydrate
- SKU
- R0123
Category: API's & Intermediates
- Synonyms
- LEA 103 , Naropin , (S)-Ropivacaine hydrochloride monohydrate
- 132112-35-7
- CAS-Number
- C17H26N2O · HCl · H2O
- Molecular Formula
- 328.88
- Molecular Weight
Specifications
- Purity
- ≥98% (T)
- Appearance
- White to off-white powder
- Identity
- 1H-NMR
Properties
- Solvents
- DMSO (25mg/mL), DMF (15mg/ml), ethanol (15mg/ml), methanol, water (2mg/ml).
- Melting Point
- 267-269°C (lit.)
- Boiling Point
- 410.2° C at 760 mmHg (Predicted)
- Optical Activity
- optical purity: -6 to -10 degree
- Shipping
- AMBIENT
- Short Term Storage
- +4°C
- Long Term Storage
- +4°C
- Handling Advice
- Protect from light and moisture.
- Use / Stability
- Stable for at least 2 years after receipt when stored at +4°C.
- Hazard statements
- H318
- Precautionary statements
- P280, P305 + P351 + P338, P310
- GHS Symbol
- GHS05
- Signal word
- Danger
- Transportation
- Not dangerous goods
- Description
- Ropivacaine is a long-acting amide local anaesthetic agent. Ropivacaine is a potent and reversible blocker of sodium channels in nerve fibers. It is a pure S(-)enantiomer and less lipophilic than racemic sodium channel blockers, such as bupivacaine and is less likely to penetrate large myelinated motor fibres, resulting in a relatively reduced motor blockade. Thus, ropivacaine has a greater degree of motor sensory differentiation, which could be useful when motor blockade is undesirable. The reduced lipophilicity is also associated with decreased potential for central nervous system toxicity and cardiotoxicity.
- Smiles
- CC1=CC=CC(C)=C1NC(C2CCCCN2CCC)=O.O.Cl
- InChi Key
- VSHFRHVKMYGBJL-UHFFFAOYSA-N
- References
- (1) R.D. Wang, et al., Exp. Opin. Pharmacother. 2, 2051 (2001) (Review) , (2) T.G. Hansen, Exp. Rev. Neurother. 4, 781 (2004) (Review) , (3) G. Kuthiala & G. Chaudhary, Indian J. Anaesth. 55, 104 (2011) (Review)
Ropivacaine is a long-acting amide local anaesthetic agent. Ropivacaine is a potent and reversible blocker of sodium channels in nerve fibers. It is a pure S(-)enantiomer and less lipophilic than racemic sodium channel blockers, such as bupivacaine and is less likely to penetrate large myelinated motor fibres, resulting in a relatively reduced motor blockade. Thus, ropivacaine has a greater degree of motor sensory differentiation, which could be useful when motor blockade is undesirable. The reduced lipophilicity is also associated with decreased potential for central nervous system toxicity and cardiotoxicity.