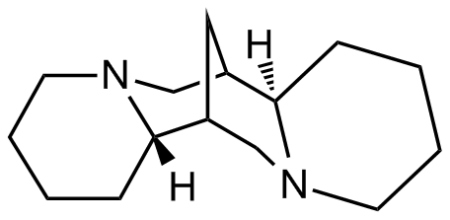
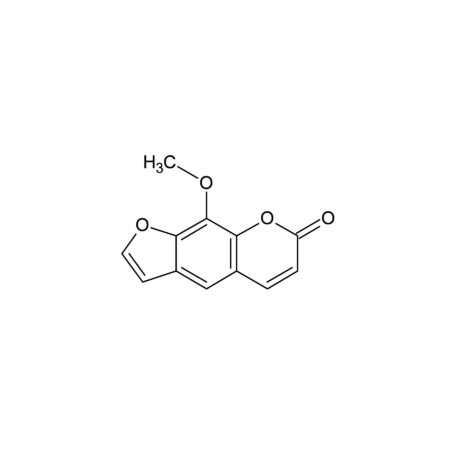
(+)-Sparteine
- SKU
- S0048
Category: Phytochemicals
- Synonyms
- Pachycarpine
- 492-08-0
- CAS-Number
- C15H26N2
- Molecular Formula
- 234.38
- Molecular Weight
Specifications
- Purity
- ≥98% (NMR)
- Appearance
- Colourless to light yellow liquid
- Identity
- 1H-NMR
Properties
- Solvents
- ethanol, water (3 mg/ml)
- Melting Point
- 201 °C
- Boiling Point
- 174 °C (8mmHg, lit.)
- UV / Vis
- λmax 202nm (CH3CN) (lit.)
- Optical Activity
- [α]/D +17.0±1.5°, c = 1 in ethanol
- Density
- 1.020 g/cm3
- Shipping
- AMBIENT
- Short Term Storage
- +4°C
- Long Term Storage
- +4°C
- Handling Advice
- Protect from light and air.
- Use / Stability
- Stable for at least 2 years after receipt when stored at +4°C.
- Hazard statements
- H302 + H312 + H332
- Precautionary statements
- P261, P264, P270, P280, P301 + P312 + P330, P302 + P352 + P312, P304 + P340 + P312, P363, P501
- GHS Symbol
- GHS07
- Signal word
- Warning
- Transportation
- Not dangerous goods
- Description
- (+)-Sparteine is a class 1a antiarrhythmic agent sodium channel blocker. It is an alkaloid and can be extracted from scotch broom. It is the predominant alkaloid in Lupinus mutabilis, and is thought to chelate the bivalent cations calcium and magnesium. As a building block and intermediate it is used as an amine chiral ligand in organic chemistry, especially in syntheses involving organolithium reagents.
- Smiles
- [H][C@]12N(CCCC2)C[C@@H]3[C@@]4([H])N(CCCC4)C[C@H]1C3
- InChi Key
- SLRCCWJSBJZJBV-TUVASFSCSA-N
- References
- (1) S. Voitenko, et al., Mol. Pharmacol. 40, 180 (1991) , (2) H. Helmke & D. Hoppe, Synlett 9, 978 (1995) , (3) A.R. Germain, et al., J. Org. Chem. 76, 2577 (2011) , (4) G. Vulugundam, et al., Chem. Commun. 52, 7513 (2016)
- InChi
- InChI=1S/C15H26N2/c1-3-7-16-11-13-9-12(14(16)5-1)10-17-8-4-2-6-15(13)17/h12-15H,1-11H2/t12-,13-,14-,15+/m1/s1
(+)-Sparteine is a class 1a antiarrhythmic agent sodium channel blocker. It is an alkaloid and can be extracted from scotch broom. It is the predominant alkaloid in Lupinus mutabilis, and is thought to chelate the bivalent cations calcium and magnesium. As a building block and intermediate it is used as an amine chiral ligand in organic chemistry, especially in syntheses involving organolithium reagents.