Specifications
- Purity
- ≥98% (HPLC)
- Appearance
- Faint yellow to yellow powder
- Identity
- 1H-NMR
Properties
- Solvents
- acetone
Category: API's & Intermediates
Anthralin is a leukotriene biosynthesis inhibitor. It inhibits LTB4 omega-oxidation and disrupts mitochondria function. Anthralin is used in the treatment of psoriasis, as a fungicide, in the treatment of ringworm infections and in chronic dermatoses. It accumulates in mitochondria where it interferes with the supply of energy to the cell, probably by the oxidation of dithranol releasing free radicals. This impedes DNA replication and so slows the excessive cell division that occurs in psoriatic plaques. Numerous studies have demonstrated anti-proliferative and anti-inflammatory effects of anthralin on psoriatic and normal skin. It is also used as matrix substance for MALDI-MS (matrix-assisted laser desorption ionization MS).
| Synonyms | Dithranol, 1,8-Dihydroxyanthrone, Anthracene-1,8,9-triol, NSC 43970, NSC 629313 |
|---|---|
| Purity | ≥98% (HPLC) |
| Appearance | Faint yellow to yellow powder |
| CAS-Number | 1143-38-0 |
| Molecular Formula | C14H10O3 |
| Molecular Weight | 226.23 |
| Identity | 1H-NMR |
| Solvents | acetone |
| Smiles | OC1=CC=CC2=C1C(=O)C1=C(O)C=CC=C1C2 |
| InChi Key | NUZWLKWWNNJHPT-UHFFFAOYSA-N |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light and moisture. |
| Use / Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Hazard statements | H315-H319-H335 |
| Precautionary statements | P261-P305 + P351 + P338 |
| GHS Symbol | GHS07 |
| Signal word | Warning |
| Transportation | Not dangerous goods |
| References | (1) R.E. Ashton, et al.: J. Am. Acad. Dermatol. 9, 173 (1983), (2) J.M. Schröder: J. Invest. Dermatol. 87, 624 (1986), (3) C.E. Orfanos, et al.: Drugs 34, 459 (1987) (Review), (4) L. Kemeny, et al.: Skin Pharmacol. 3, 1 (1990), (5) K. Müller: Biochem. Pharmacol. 53, 1215 (1997) (Review), (6) K. Pavithran: Indian J. Dermatol. Venereol. Leprol. 67, 104 (2001), (7) C.H. Le, et al.: Anal. Chem. 84, 8391 (2012), (8) C. Ronpirin & T. Tencomnao: Genet. Mol. Res. 11, 412 (2012), (9) S.E. George, et al.: J. Pharm. Pharmacol. 65, 552 (2013) |
| InChi | InChI=1S/C14H10O3/c15-10-5-1-3-8-7-9-4-2-6-11(16)13(9)14(17)12(8)10/h1-6,15-16H,7H2 |
| Quantity | 1 g, 5 g, Bulk |
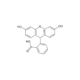
 2,4-Difluororesorcinol
2,4-Difluororesorcinol