Specifications
- Purity
- ≥99% (HPLC)
- Appearance
- White to light yellow powder
- Identity
- 1H-NMR
Properties
- Solvents
- 01 N HCl (10 mg/ml) Slightly DMSO (1 mg/ml)
Category: Antibiotics
“Ganciclovir is antiviral synthetic analog of the nucleoside 2′-deoxy-guanosine, which is used to treat or prevent cytomegalovirus (CMV) infections. It acts as a prodrug activated by phosphorylation. It is first phosphorylated to ganciclovir monophosphate by a viral kinase encoded by the cytomegalovirus (CMV) gene UL97 during infection. Subsequently, cellular kinases catalyze the formation of ganciclovir diphosphate and ganciclovir triphosphate. Ganciclovir triphosphate is a competitive inhibitor of deoxyguanosine triphosphate (dGTP) incorporation into DNA and preferentially inhibits viral DNA polymerases more than cellular DNA polymerases. It is also useful in the study of gene therapy in cancer research. Ganciclovir has been used in the study of “”suicide”” gene therapy in cancer research, where upon expression of a viral suicide gene encoding herpes simplex virus, thymidine kinase (TK), the non-toxic prodrug ganciclovir, is converted to an active phosphorylated analog that can be incorporated into the DNA of replicating eukaryotic cells, causing death of the malignant dividing cell. Causes an irreversible cell cycle arrest at the G2/M checkpoint. Ganciclovir has also been used to study the loss of telomeres and to evaluate sensitivity of viruses to antiviral agents.”
| Synonyms | 2-Amino-1,9-dihydro-9-[[2-hydroxy-1-(hydroxymethyl)ethoxy]methyl]-6H-purin-6-one, 9-(1,3-Dihydroxy-2-propoxymethyl)guanine, DHPG, BW 759, 2'-Nor-2'-deoxyguanosine |
|---|---|
| Purity | ≥99% (HPLC) |
| Appearance | White to light yellow powder |
| CAS-Number | 82410-32-0 |
| Molecular Formula | C9H13N5O4 |
| Molecular Weight | 255.23 |
| Identity | 1H-NMR |
| Solvents | 01 N HCl (10 mg/ml) Slightly DMSO (1 mg/ml) |
| Smiles | O=C1C2=C(N(COC(CO)CO)C=N2)N=C(N)N1 |
| InChi Key | IRSCQMHQWWYFCW-UHFFFAOYSA-N |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Handling Advice | Protect from light and moisture. |
| Use / Stability | Stable for at least 2 years after receipt when stored at +4°C. |
| Hazard statements | H340-H361fd |
| Precautionary statements | P201-P202-P280-P308 + P313-P405-P501 |
| GHS Symbol | GHS08 |
| Signal word | Danger |
| Transportation | Not dangerous goods |
| References | (1) T. Matthews & R. Boehme, Rev. Infect. Dis. 10, S490 (1988) (Review), (2) S. Paul & S. Dummer, Am. J. Med. Sci. 304, 272 (1992) (Review), (3) P.J. Halloran & R.G. Fenton, Cancer Res. 58, 3855 (1998), (4) J. Qiao, et al., Hum. Gene Ther. 11, 1569 (2000), (5) M. Aghi, et al., J. Gene Med. 2, 148 (2000) (Review), (6) C. Fillat, et al., Curr. Gene Ther. 3, 13 (2003) (Review) |
| InChi | InChI=1S/C9H13N5O4/c10-9-12-7-6(8(17)13-9)11-3-14(7)4-18-5(1-15)2-16/h3,5,15-16H,1-2,4H2,(H3,10,12,13,17) |
| Quantity | 100 mg, Bulk |
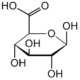
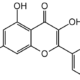 Galangin
Galangin